|
The outermost shell also called the valence shell, which contains the valence electrons of the atom. 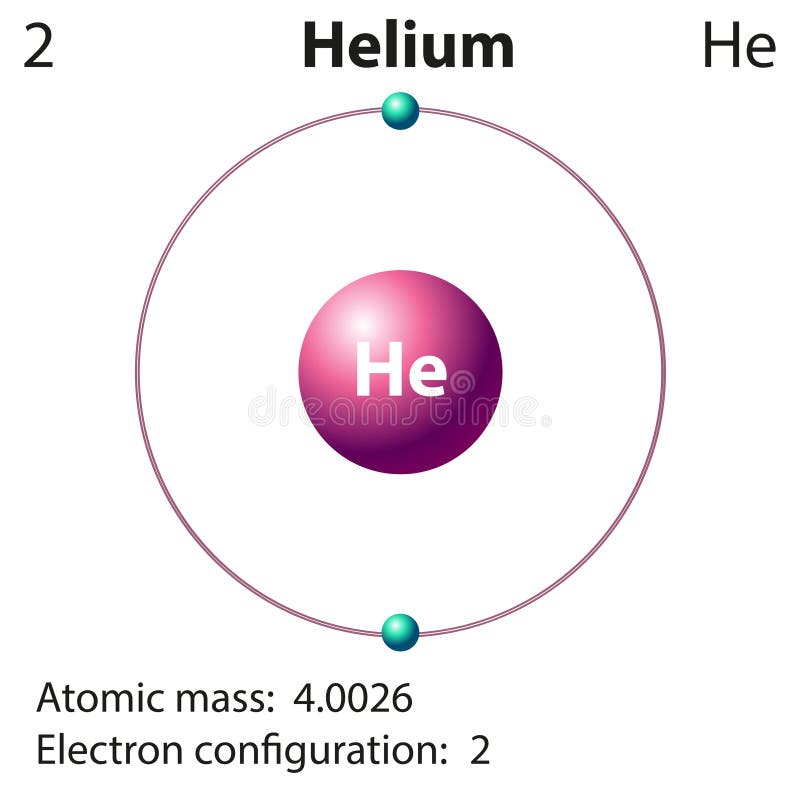
What is the outer shell of the Bohr diagram of the Helium atom? So, the Helium atom belongs to the 1st Period in the periodic table, hence, the number of electron shells for the Bohr model of Helium will be only 1 (K-shell). The elements or atoms in the first period of the periodic table have one energy level or one electron shell, same as, the elements in the second period have two energy levels or two-electron shells, and so on… It only has two electrons in its outer shell so its valence electron configuration is 1s2. How to find the electron configuration using different methods?įAQ How many electron shells a Helium Bohr model contains?Įlectron shells also called energy levels, you can find the number of electron shells for an element by knowing its period number in the periodic table. Helium is slightly different than the other noble gas elements.Or the electronic configuration of Helium is 1s 2since it contains a total of 2 electrons and these two electrons will go in a 1s orbital. Helium has both of its electrons in the 1s orbital (1s2). So, based on the shell, the electronic configuration of the Helium atom is. Hydrogen has one electron in a 1s orbital and we write its electron configuration as 1s1. The K-shell or outer shell of the Helium Bohr model contains 2 electrons, therefore, the number of valence electrons in the Helium atom is 2. Hence, the electrons found in the K-shell of the Helium atom are its valence electrons because it is the outermost shell also called the valence shell. So, we have to find a valence electron in the Helium atom, for this, look at its Bohr diagram.īohr’s diagram of Helium has only one electron shell (K-shell ). This means it will not react with other atoms. The electron configuration for Helium shows a full outer shell and is Helium is therefore called a Nobel Gas. Because the 1s orbital is full with 2 electrons and any additional electrons would go in a new energy level. These electrons have more energy compare to the inner shell electrons.įrom the Bohr diagram of an atom, we can easily find the number of valence electrons in an atom by looking at its outermost shell. Helium only has 2 electrons and therefore it has a configuration of 1s 2. Valence electrons are found in the outermost shell of an atom and they can take participate in the formation of a chemical bond. Find the Valence electron of Helium through its Bohr diagram
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
